Computer System Validation (CSV) V/S Computer Software Assurance (CSA) – Change is On the Way
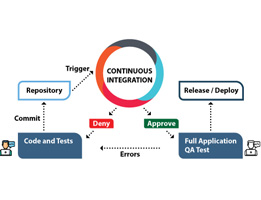
The FDA’s new approach to CSV is Computer Software Assurance (CSA). It represents a step-change in computer system validation, placing critical thinking at the centre of the CSV process. CSA is a risk- based approach to validate a system directly impacting Product Quality and Patient Safety. The traditional approach to…