The FDA’s new approach to CSV is Computer Software Assurance (CSA). It represents a step-change in computer system validation, placing critical thinking at the centre of the CSV process. CSA is a risk- based approach to validate a system directly impacting Product Quality and Patient Safety. The traditional approach to Computer System Validation (CSV) has […]
Pharmaceutical manufacturing is one of the most closed regulated industry. The Pharmaceutical industry have last 50 years of admirable quality experience. Vetting of Quality Assurance is perspective meets its regulatory specifications. Compliance can’t take granted if their products are not meeting to their appreciate safety, Quality, requirements, customers satisfactions. Quality control is the part of […]
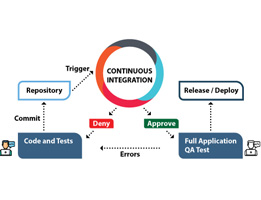
Continuous integration (CI) is a method of integrating changes needed in one single project which is developed, then tested accordingly, this method helps developers to deliver a software in a more efficient and a predictable manner. To understand the importance of continuous integration first we understand how the process is going before continuous integration is […]
Each regulated record (is a GMP record) and should be periodically reviewed Strong SOP and Work instructions repository covering all working areas Templates for all documentation, processes – recommended, mandatory VMP for computer systems – mandatory for all who are regulatory audited and use the same for validation across for computer systems URS – always […]
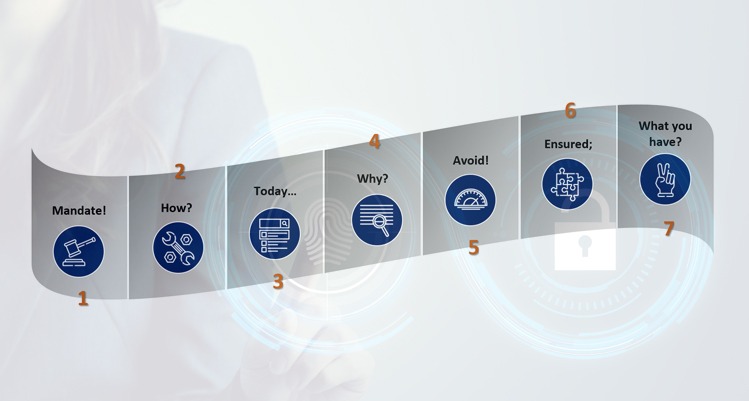
CSV is mandatory for implementation of software systems in a GxP regulated environment. And, it would incur huge costs if not handled adequately. With increasing number of software systems implemented in any organization, it becomes utmost important to check the costs at every step, especially of CSV. The CSV tools include the following: Mandate! To […]
Computer System Validation (CSV) is the practice of ensuring that any software or hardware which meets its purpose with regulatory guidelines. The pharmaceutical business model is emerging, and CSV is becoming even more crucial because of the increasing scope of information systems, more integrated functionality, consolidation, and archiving requirements. It helps companies to maintain its […]
Very “synonymous words”, DS and DQ, and can many a time be misleading, We might want to say one but speak the other and then would need clarifying. One of the set of such words include – Design Specification (DS) and Design Qualification (DQ).
Qualification – applies to hardware, devices; not the process in which the hardware is used. Thus, verifies the hardware is suitable to do a task. Verification – applies to the evaluation at intermediate steps when the software is developed to check for the correct approach to the final product.
A query… It is a common belief across that the softwares developed by known and established software vendors (with a brand value) are more accepted, preferred by the companies. And, also they face less queries and questions; are more accepted from the regulatory agency auditors than the other establishing software vendors. Is this true?
In the last few years, cloud hosting has emerged as a preferred choice which has enabled companies to get out of maintaining their own servers to host various software applications where cloud computing is a model for enabling 24×7, convenient, on-demand network access to a shared pool of configurable computing resources (e.g., networks, servers, storage, […]
- 1
- 2