
A risk-based enterprise quality management system
An industry-ready solution for pharma and life sciences that handles all your quality challenges, ensuring full compliance with configurable workflows and comprehensive audit trails.
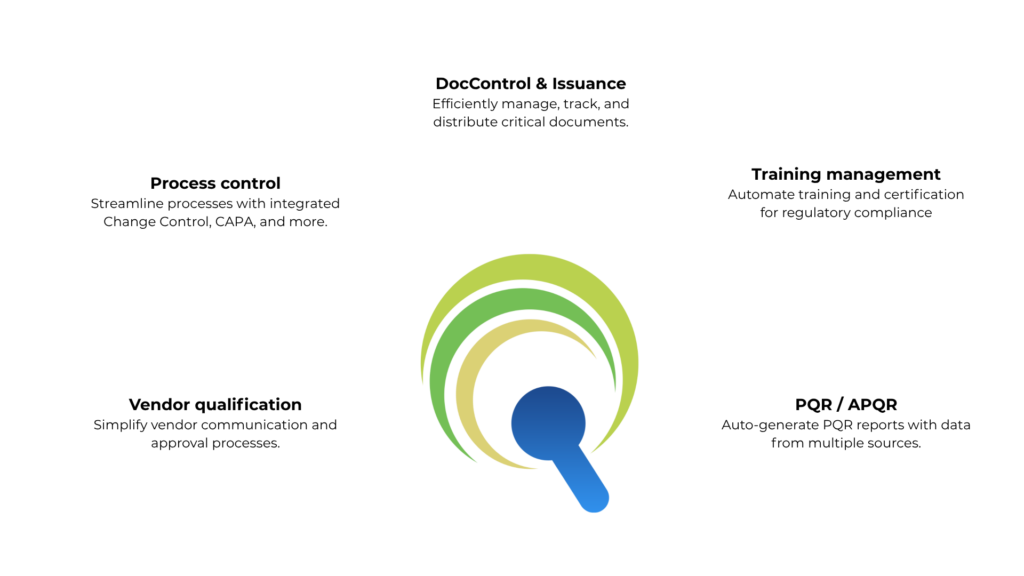
QEdge is an integrated Quality Management System (QMS) tailored for pharmaceutical manufacturing and life sciences industries. Designed to meet stringent regulatory standards like FDA, EU-GMP, and ICH guidelines, QEdge streamlines quality processes across Change Control, Deviation, CAPA, Document Management, Training, and Vendor Qualification. With features like SOP lifecycle management, SCORM-based eLearning, and audit-ready compliance tracking, QEdge ensures operational excellence, data integrity, and regulatory readiness for GxP environments.
What makes Quality Management Sytem by Sarjen Systems stand out?
Impact
Implementations
Users
Quality processes
Documents managed
SOP's implemented
Trainers qualified
PQR submissions
Vendors qualified
Features
Always audit-ready
Easy process versioning
Configurable processes
FDA and EU-compliant workflows
Field and functional audit trails
User friendly

