
Smarter dossier management for faster approvals
Our global dossier publishing platform simplifies dossier management and submission. By automating workflows for regions and products, it ensures a consistent and efficient process, reducing complexity and improving accuracy.
Using our global dossier management and publishing platform helps standardise and streamline your regulatory submission process. With automated workflows tailored to specific regions, products, and regulatory authorities, it accelerates dossier creation, validation, and submission. This results in fewer compliance issues, reduced manual errors, and faster regulatory approvals. Our solution increases operational efficiency, ensures data consistency, and supports audit-ready documentation—saving time, improving accuracy, and keeping your submissions compliant so you can focus on scaling your business.
What makes eCTD/CTD/NeeS Publishing solution by Sarjen Systems stand out?
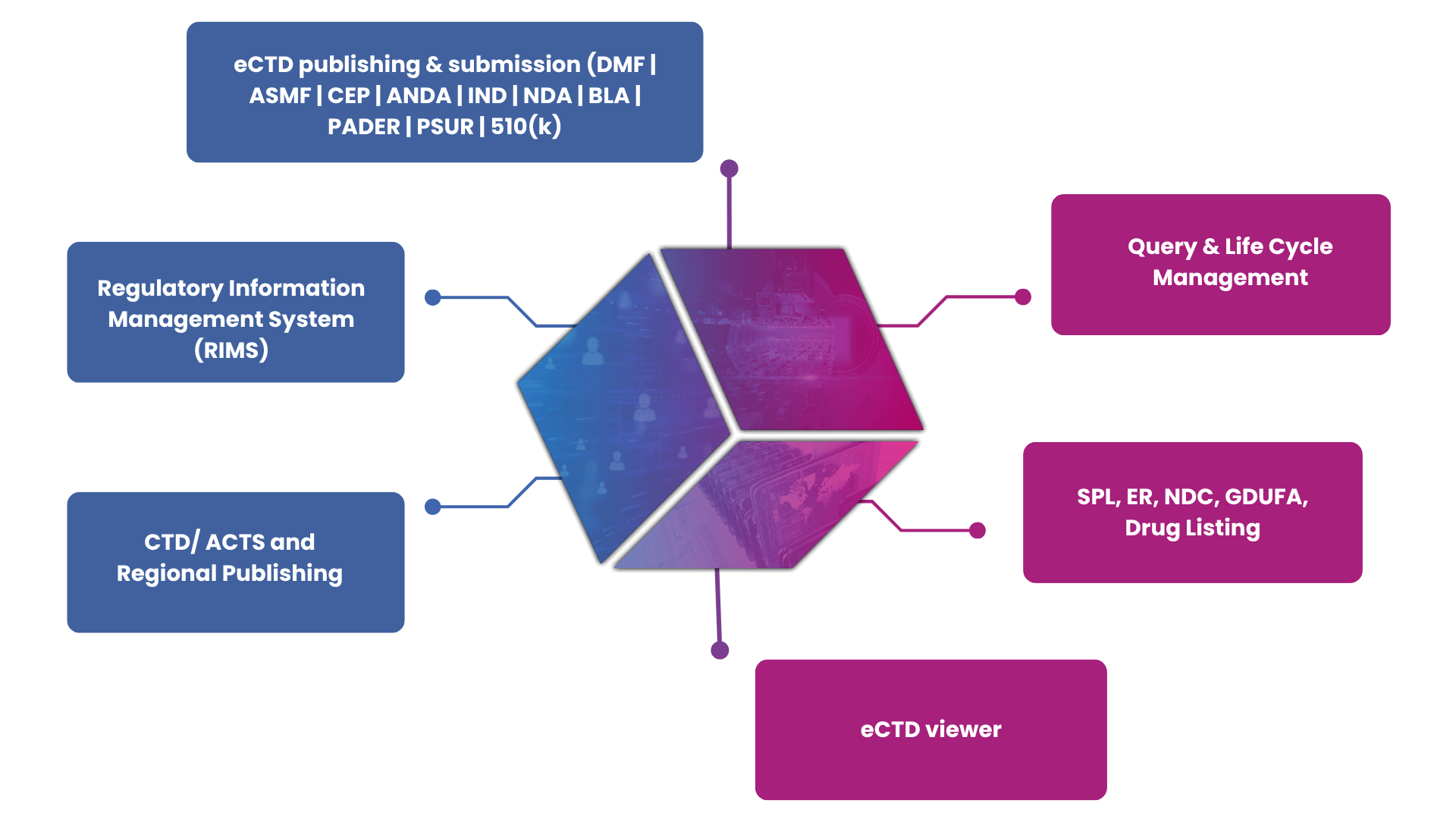
Impact
Country structures
Semi-regulated markets submissions
Dossiers
Non-regulated markets submissions
SPLs
Features
Region-specific Dossier Management
Product-based Dossier Lifecycle
Automated submission workflow
Regulatory compliance support
Real-time document tracking
Version control
Easy Dossier review & approval
Boosted ROI and accuracy

