
A unified RIMS for global compliance
Our Regulatory Information Management System (RIMS) simplifies regulatory processes through a centralised, IDMP-compliant platform with intuitive workflows for efficient data tracking and global compliance.
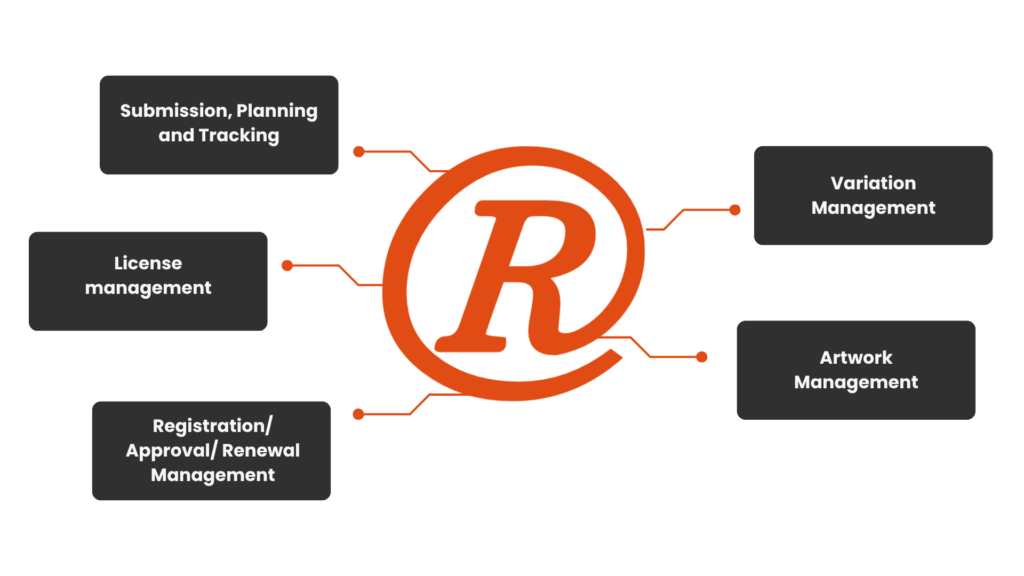
Implementing RIMS gives you better control and visibility over your regulatory processes, significantly reducing manual errors and time spent on compliance tasks. By centralising all regulatory data, you can easily track submissions, approvals, variations, and renewals, improving overall efficiency. RIMS helps your team stay compliant with global regulations, streamlining product lifecycle management from registration to approval, and enhancing your ability to make data-driven decisions. The result? Faster go-to-market timelines and reduced operational costs.
What makes Early-phase clinical trials by Sarjen stand out?
Features
IDMP compliant
Pre-Validated for faster implementation
Enhanced compliance management
Robust audit trail
Centralised data for improved decision-making

